What You’ll Learn In This Post
- How cloud droplets form via condensation nuclei
- How saturation works
- How forest fires can create clouds
- Four ways that air can be lifted to form clouds
Introduction
Now that we know the fundamentals of the phase changes of water, we’re going to turn our attention to the most readily identifiable process resulting from those phase changes: cloud formation. Clouds contain tiny liquid droplets and/or ice crystals, which collectively we call “hydrometeors”. But what ingredients are needed to form these hydrometeors out of thin air?
Table of Contents

Cloud formation by saturation
What’s a hydrometeor? (Click to expand)
A hydrometeor (or “precipitation particle”) is an individual water droplet or ice crystal suspended in the air. Sometimes, the term “hydrometeor” may refer to only those hydrometeors that are large enough to fall towards the ground. A hydrometeor differs from a water molecule in that it is MUCH bigger than an individual water molecule and contains MANY, MANY water molecules. For example, one 0.5 mm diameter drizzle drop contains over one quadrillion water molecules!
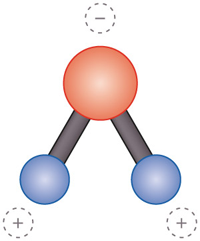
Water molecules consist of one oxygen atom and two hydrogen atoms (see image above). Water molecules are bipolar molecules, meaning that different ends of the molecule have opposite charges. Being larger, the oxygen atom tends to attract electrons and carries a negative charge. Meanwhile, the hydrogen atoms at the opposite end of the water molecule tend to be positively charged.
When a high concentration of water molecules in a very small region of the atmosphere brings those molecules closely together, they begin to form weak bonds between each other known as hydrogen bonds (shown below). Hydrogen bonds connect oppositely-charged ends of neighboring water molecules into a loose connection that links them together. The growth of these small clusters of water molecules into larger droplets is aided by small bits of dust and other hydrophilic (water-friendly) particles (“aerosols”), known as condensation nuclei. Not all aerosols are condensation nuclei, but all condensation nuclei can form a droplet.
At colder temperatures, ice nuclei are needed for ice crystals to form. More details on this nucleation process are provided in the Microphysics chapter. Regardless of whether droplets or ice crystals are produced, we call the air “saturated” when water molecules accumulate around the condensation nucleus and begin to form small droplets in the net phase change of condensation. The growth of many large droplets produces cloud formation.
Why did the air molecule break up with its partner? (Click to expand)
Because it said, “I need some space! I’m completely saturated with moisture, and there’s no room left for any more water vapor!”
When the air is saturated, the water molecules in the air have developed an equilibrium between condensation (water molecules accumulating on the hydrometeor) and evaporation (water molecules leaving the hydrometeor). At saturation, an approximately equal number of molecules are joining a water droplet as those that are leaving it. This is also indicated by the air temperature and dewpoint temperature: they are equal. We’ll expand upon the concepts of saturation and dewpoint temperature in a future post.
Fundamentally, saturation involves the equilibrium, or balance, between different phase changes of water. Water vapor is naturally held in the air. Too much water vapor in one place causes some of that water vapor to change phase, forming either liquid droplets or ice crystals. Too little water vapor in one place, and any existing liquid droplets or ice crystals will lose some of their water molecules to the vapor phase, and they may evaporate completely. To form a cloud, there must be a large amount of excess water vapor (saturated or supersaturated air) in a small portion of the atmosphere.
What is needed to form a cloud? (Click for answer)
Cloud formation requires two key ingredients: saturation and condensation nuclei.
In the video below, you can see a pyrocumulus cloud growing from a forest fire in southern California. The smoke from the fire provides soot particles that serve as condensation nuclei, which are activated (moistened to form growing droplets), forming cloud droplets when mixed with the surrounding air. The soot is propelled upward by the intense heat of the forest fire, which creates instability in the lower atmosphere. This instability produces rising motion which propels the cloud upward, as we’ll see in the Stability chapter.
The pure white clouds you see growing and dissipating at higher altitudes are composed of water droplets. You’ll notice how the clouds are “dirty” below a certain level and white above that level: that separating layer is the “cap”, a thin, warm layer which acts as a barrier to the rising soot particles in the air.
Clouds form in areas of rising air
We know from the last post that clouds form in regions of moist, rising air, but how does that air begin to rise in the first place? In the video above, the lifting mechanism is the extreme surface heating produced by the forest fire. It doesn’t take that much heat to produce clouds, though.
On warm, sunny days, heating of the ground by the intensity of the sun’s rays produces rising “thermals” of buoyant, warm air, which rise and cool to generate fair-weather cumulus clouds that can grow into larger clouds under the right conditions. Besides this surface heating process, there are three other common lifting mechanisms that can produce low-level rising air, as you can see on the slide below. (A chunk of air that rises or sinks in the atmosphere is known as an air parcel.)

We learned just above how surface heating contributes to cloud formation. When wind encounters a mountain range, it is forced to rise. This orographic lifting, produced by higher topography, can create clouds along mountain slopes.
Because cold air is denser than warm air, a front – that is, a boundary between warm and cold air – naturally lifts the less dense, warm air over the more dense, colder air.
And finally, when low-level air converges (comes together from different directions), this can produce lift, as the converging air forces the air to rise.
In this post, we learned about the two atmospheric ingredients necessary for cloud formation. In a future post, we’ll complete the story of how clouds form by learning how the atmosphere “bakes” these ingredients to make clouds…by cooling the air! However, I suggest that you read the next few posts in order to learn how we extend the basic concepts discussed so far to real-world clouds.
Summary: What is the Recipe for a Cloud?
Clouds are regions of concentrated liquid droplets and/or ice crystals in the atmosphere. They form thanks to two key local ingredients: saturation (a concentrated region of water vapor molecules) and condensation nuclei (tiny particles on which water vapor readily condenses). Many clouds also require low-level air to be lifted; this lifting cools the air to saturation. To produce the lift, one of four processes is required: surface heating, orographic lifting, near-surface convergence, or frontal lifting. We’ll add more layers of understanding about the cloud formation process later in the chapter.
Take the quiz below to review key concepts from this blog post.
In the next installment, we’ll look at how we name clouds.


You must be logged in to post a comment.